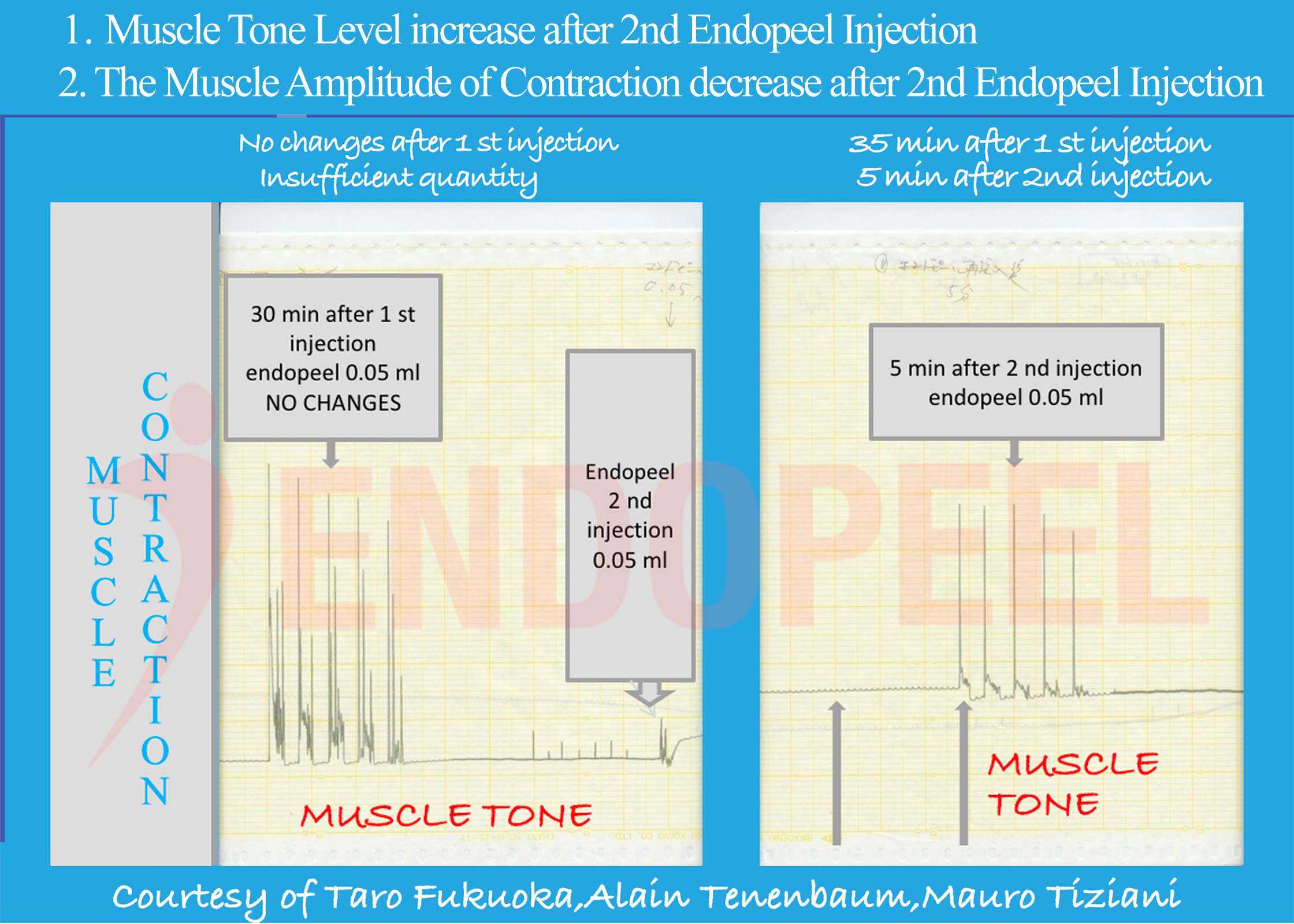
Experimental Evidence of Functional Muscle Modulation
Endopeel Physiology
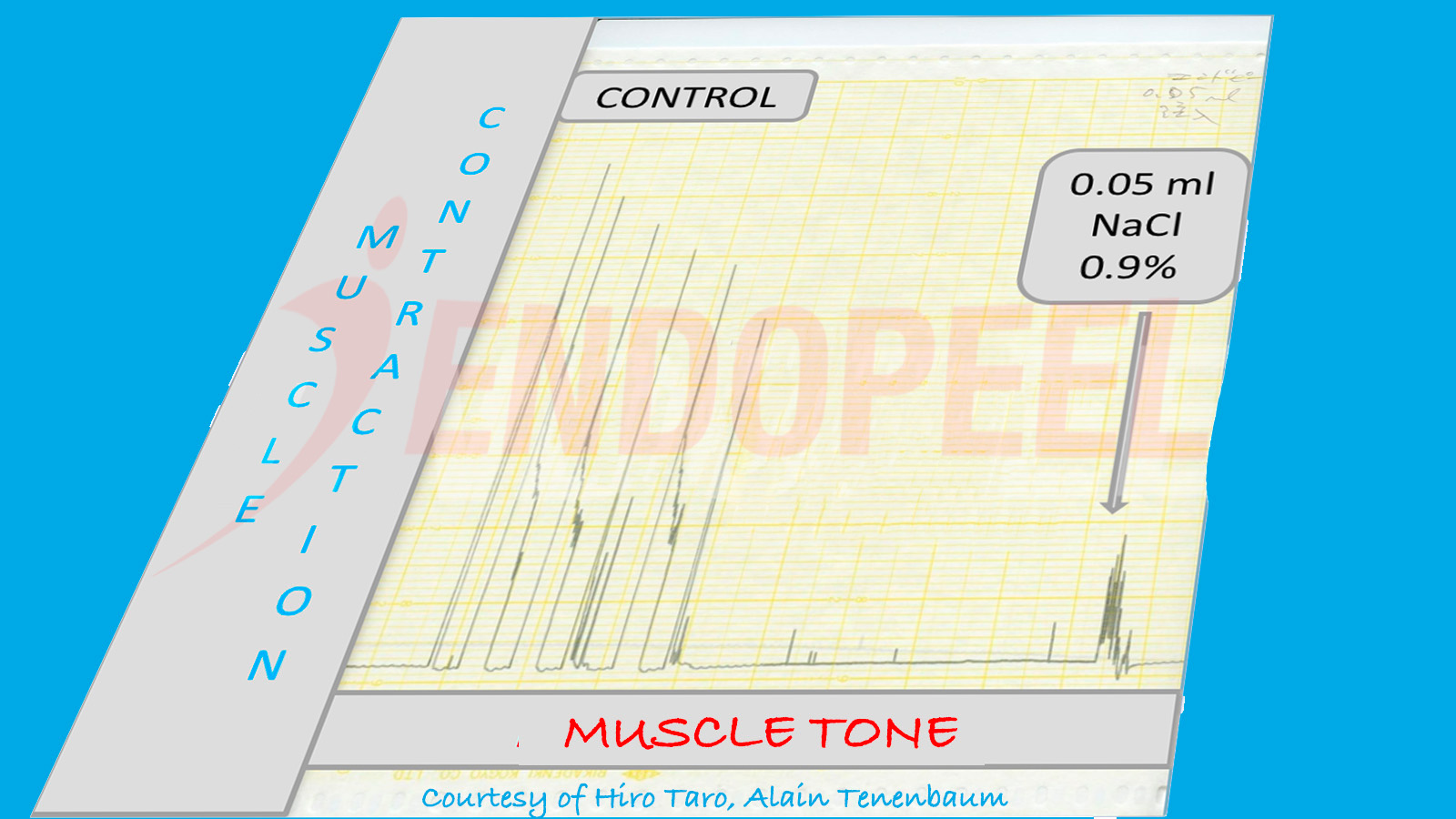
This page presents the physiologic foundation of Endopeel® based on experimental muscle contraction studies conducted at Akita University School of Medicine in collaboration with the Kyoto Pathological Research Institute (2006–2007).
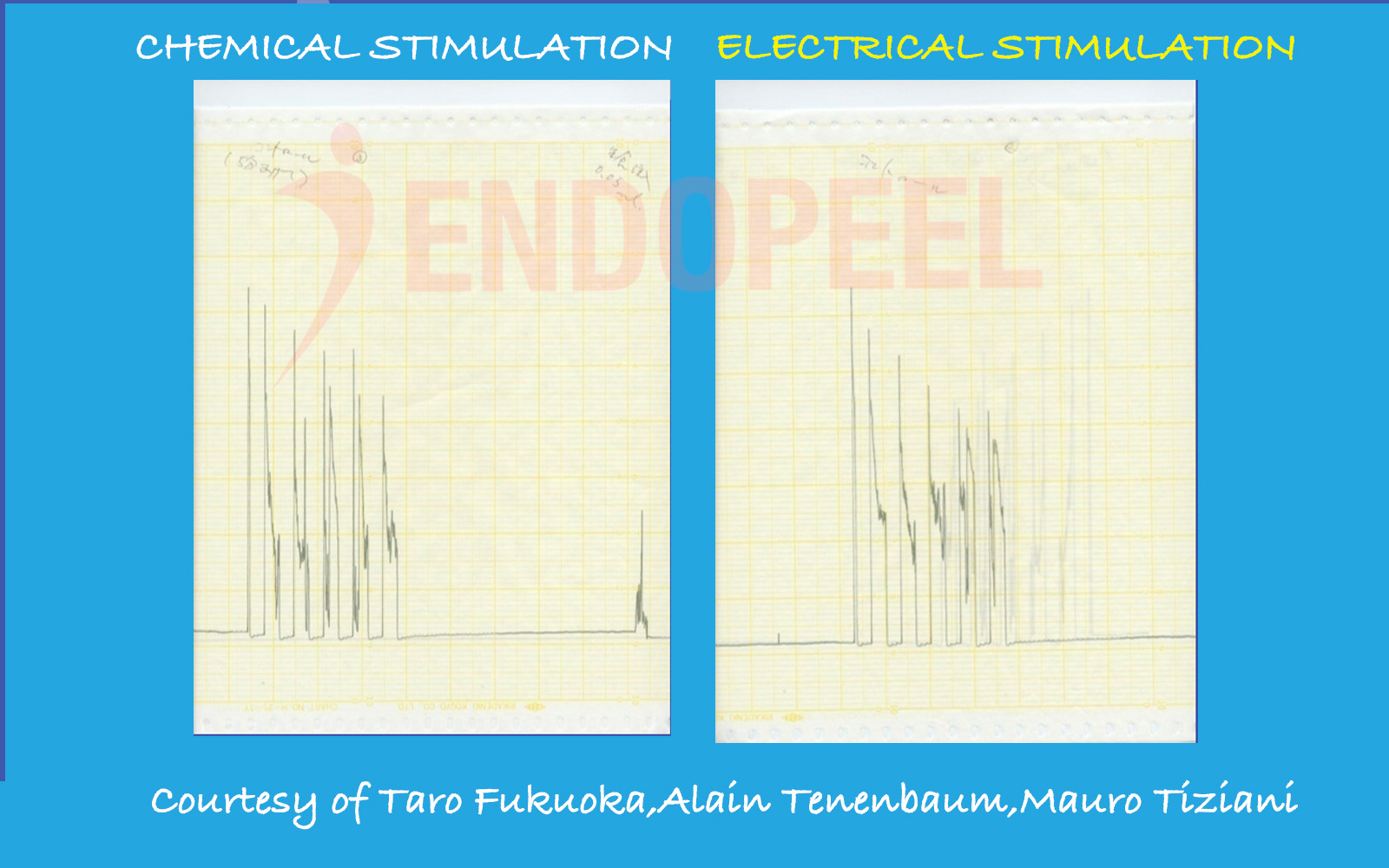
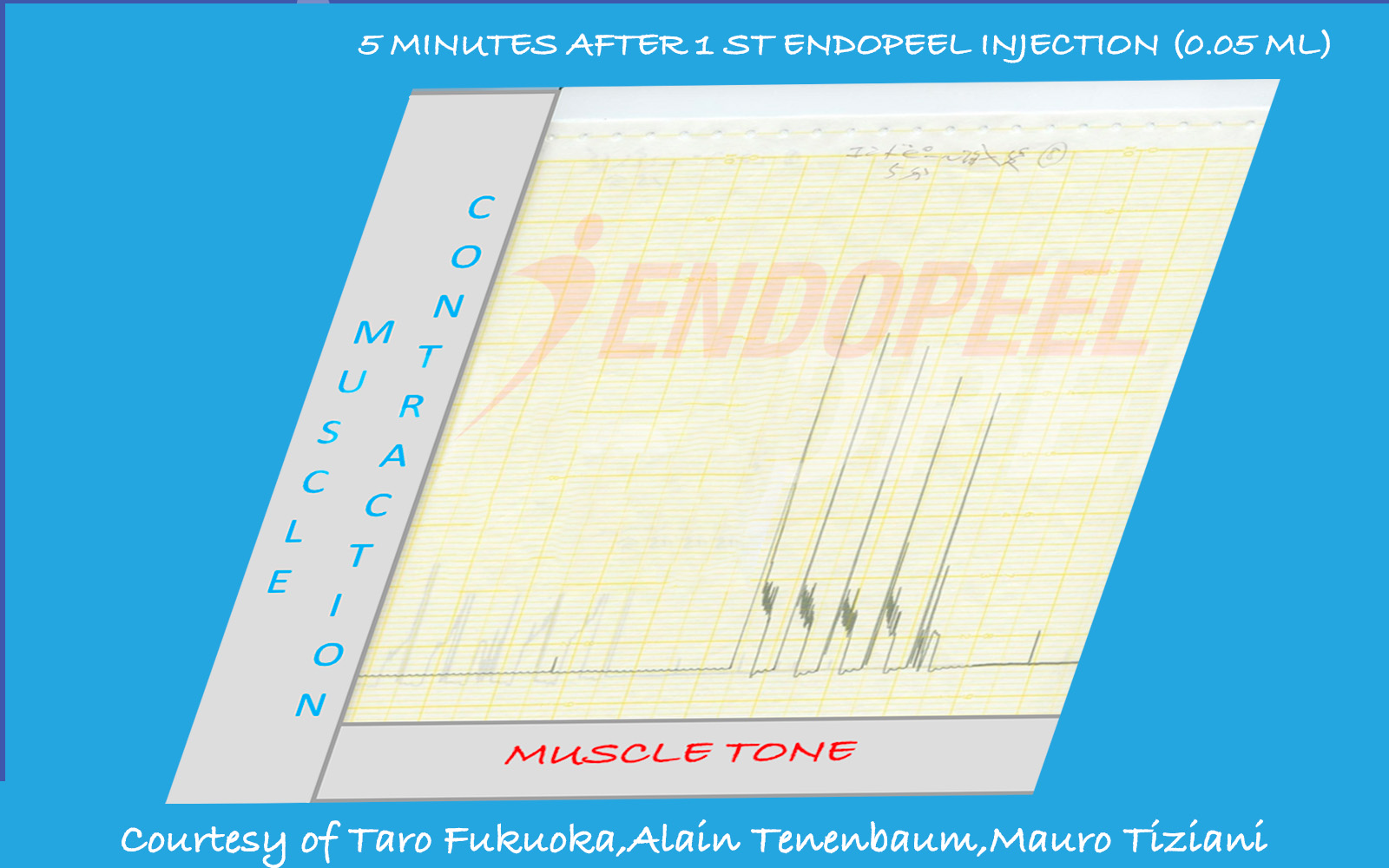
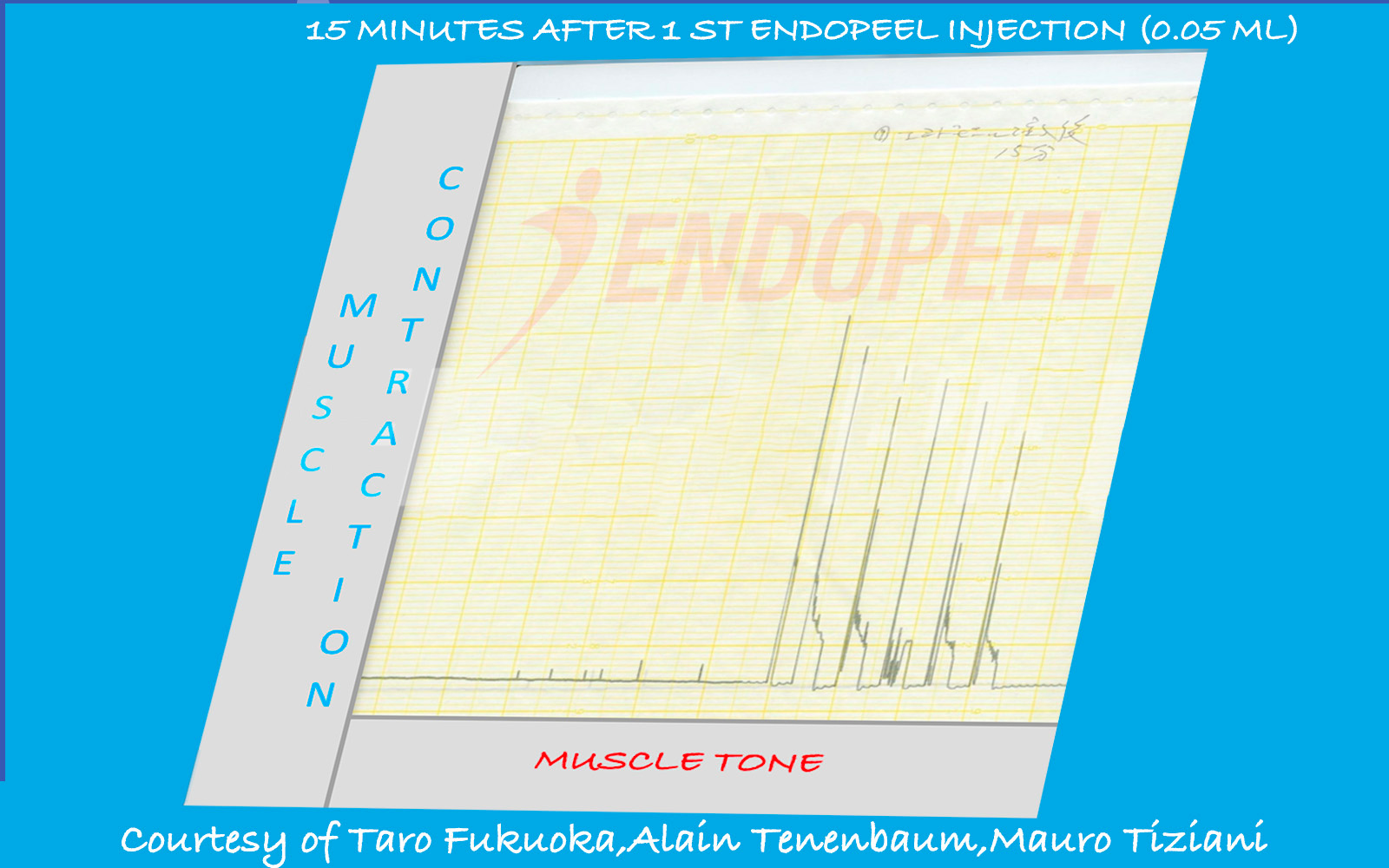
Data shown below are presented as physiologic tracings and comparative stimulation tests (electrical versus chemical), followed by the observed response after sequential Endopeel® injections.